Category: Legal Framework

- Mar, 28 2026
- Comments 1
What is an ANDA: Abbreviated New Drug Application Explained for FDA Approval
Discover what an ANDA is and how the FDA approves generic drugs. Learn the difference between ANDA and NDA, bioequivalence requirements, and the role of the Hatch-Waxman Act.
Read More
- Feb, 11 2026
- Comments 14
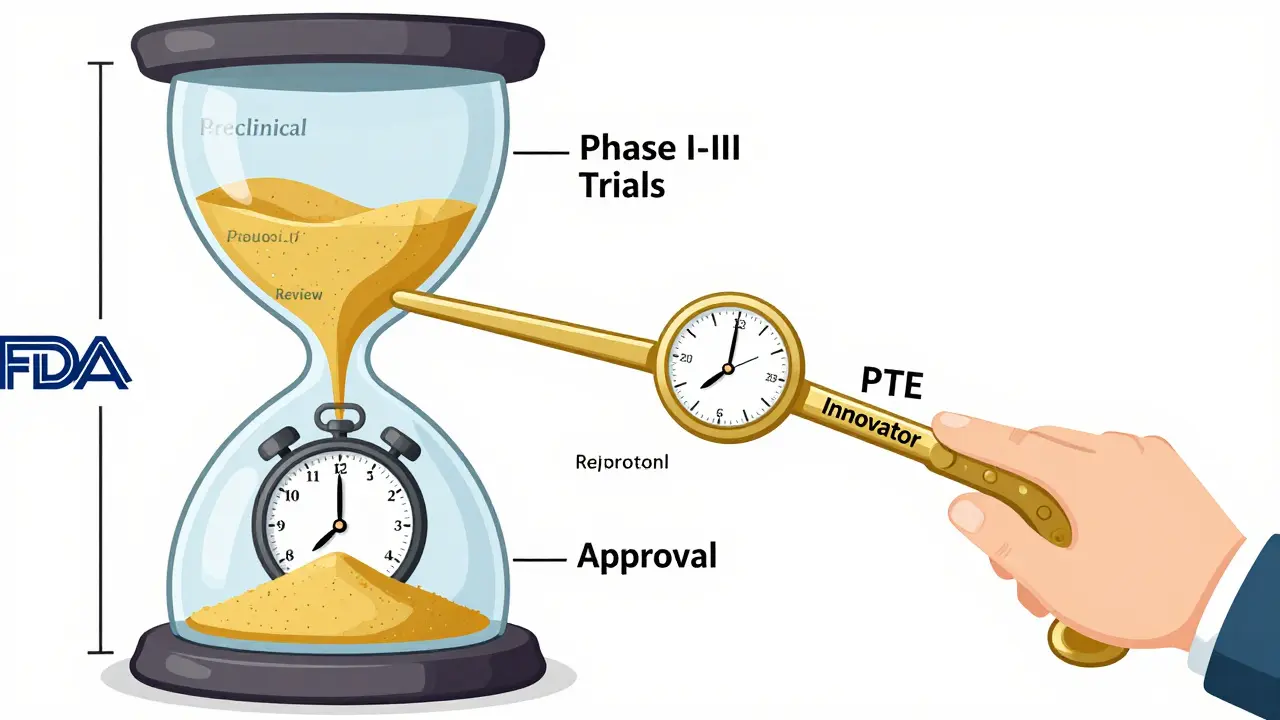
Patent Term Restoration (PTE): How It Extends Drug Patent Life
Patent Term Restoration (PTE) lets drugmakers recover lost patent time due to FDA delays. Learn how the Hatch-Waxman Act works, who qualifies, why applications get denied, and how it impacts drug prices and generic competition.
Read More

- Dec, 29 2025
- Comments 10
Liability and Indemnification in Generic Transactions: What You Need to Know
Indemnification clauses in commercial contracts determine who pays when things go wrong. Learn how liability protection works, what to look for in the fine print, and why ignoring these clauses can cost you millions.
Read More