Should You Stay on Brand NTI Drugs When Generic Substitution Is Allowed?
 Mar, 6 2026
Mar, 6 2026
When your doctor prescribes a medication like levothyroxine or warfarin, you might assume all versions are the same. But for NTI drugs-Narrow Therapeutic Index drugs-that assumption can be dangerous. These aren’t your average pills. Even tiny changes in how your body absorbs or processes them can lead to serious problems: too little, and the drug doesn’t work; too much, and it becomes toxic. The question isn’t just about cost. It’s about safety. Should you stick with the brand name, or is the generic version just as safe? The answer isn’t simple, but the data is clearer than most people think.
What Exactly Are NTI Drugs?
NTI drugs are defined by the FDA as medications where the difference between a safe, effective dose and a harmful one is razor-thin. Think of it like walking a tightrope. One step too far, and you fall. Common examples include:
- Levothyroxine (for hypothyroidism)
- Warfarin (a blood thinner)
- Tacrolimus (used after organ transplants)
- Phenytoin and carbamazepine (antiseizure meds)
These drugs aren’t rare. Millions of Americans take them daily. Levothyroxine alone is prescribed over 100 million times a year in the U.S. The FDA doesn’t publish a public list of NTI drugs, but experts agree on about 15-20 key medications that fall into this high-risk category. What makes them different isn’t the condition they treat-it’s how sensitive your body is to small changes in concentration.
How Are Generic NTI Drugs Tested?
Most generics must prove they’re bioequivalent to the brand name. That means their absorption rate in the body (measured as AUC and Cmax) must fall within 80-125% of the brand’s. For regular drugs, that’s enough. For NTI drugs? Not nearly enough.
Since 2014, the FDA has required stricter standards for NTI drugs. Instead of a flat 80-125% range, they use a "scaled" approach. Depending on the drug’s natural variability, the acceptable range can tighten to 90-111%. This sounds technical, but here’s what it means in practice: a generic levothyroxine tablet can’t vary more than 10% from the brand in how much of the drug enters your bloodstream. That’s a huge upgrade from the old standard.
Still, not all generics are created equal. The FDA assigns an "AB" rating to drugs they consider interchangeable. But even two AB-rated generics from different manufacturers can behave differently in your body. That’s because bioequivalence is tested on healthy volunteers-not patients with complex conditions. What works for a 30-year-old in a lab might not work the same for a 72-year-old with kidney disease.
Real-World Evidence: Do Generics Work?
Let’s cut through the noise. A 2022 FDA-funded study followed nearly 18,000 patients on levothyroxine over nine years. It found no meaningful difference in thyroid hormone levels between those on brand-name and generic versions. Another study of 3.5 million patients across chronic conditions like hypertension and diabetes showed identical outcomes with generics.
But here’s where it gets messy. Tacrolimus, used by transplant patients, tells a different story. Studies show that switching between generic versions-even AB-rated ones-can cause dangerous drops or spikes in blood levels. One patient might stabilize on a generic from Company A, then have a rejection episode after switching to Company B’s version. That’s why transplant centers often keep patients on the same manufacturer indefinitely.
Antiepileptic drugs are another gray zone. A 2022 survey by the Epilepsy Foundation found 42% of patients reported breakthrough seizures after switching to a generic. That’s alarming. But the study relied on self-reports, not lab tests. Still, doctors who treat epilepsy say they’ve seen it firsthand: a patient doing fine on brand-name phenytoin, then having seizures two weeks after a pharmacy switch.

Cost vs. Risk: The Real Trade-Off
Brand-name levothyroxine can cost $60 a month. The generic? $4-$15. That’s not a minor difference-it’s life-changing for people on fixed incomes. Warfarin generics save an average of $3,128 per 100 patients per year. For insurers and Medicaid, that’s billions saved annually.
But if switching costs you a hospital stay, emergency room visit, or worse, is it worth it? The answer depends on your situation. If you’re starting therapy for the first time, generics are usually fine. But if you’ve been stable on a brand for years, changing brands-even to a generic-can destabilize you. One study found that 30% of patients who switched from brand to generic levothyroxine needed dose adjustments within six months. Another 5% had severe symptoms and had to go back.
What Experts Actually Recommend
There’s no universal rule. But here’s what top medical groups agree on:
- Don’t switch unless necessary. If you’re stable on a specific product-brand or generic-stay on it. Consistency matters more than cost.
- Always check with your doctor before switching. Pharmacists can substitute automatically in many states, but for NTI drugs, that’s not always safe. Ask your doctor to write "dispense as written" on the prescription.
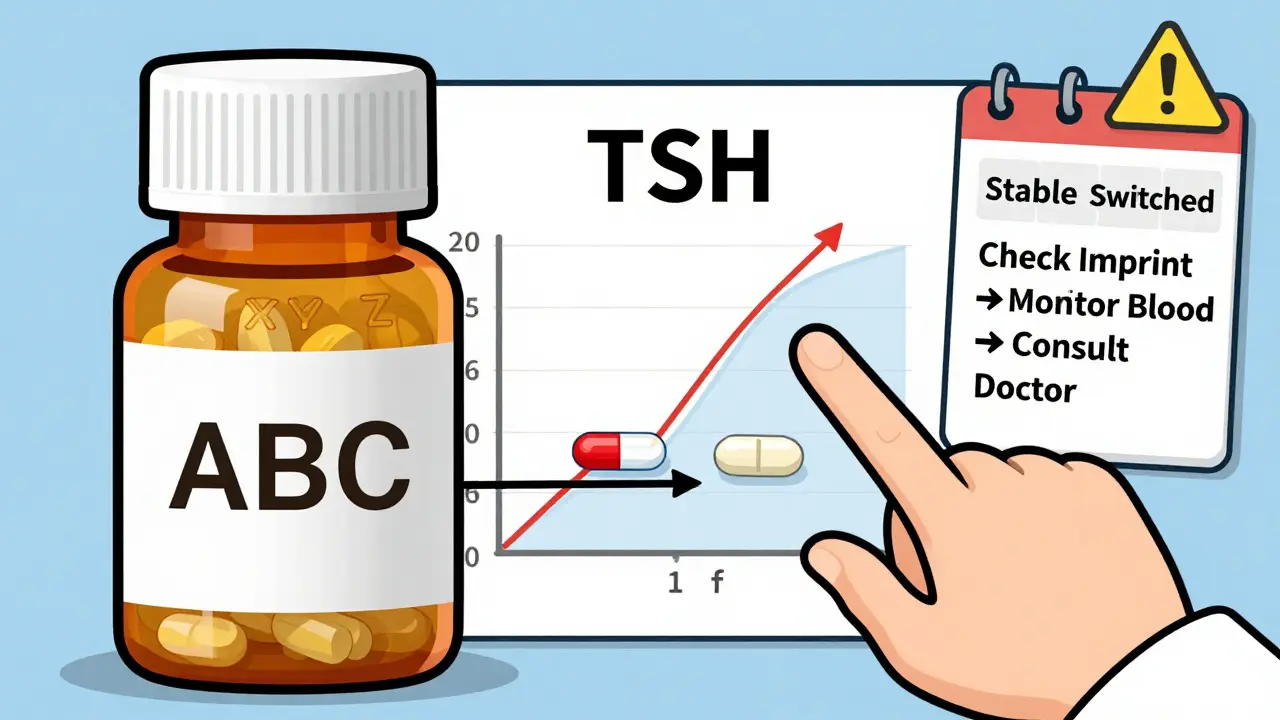
- Monitor closely after any switch. For levothyroxine, get your TSH level checked 6-8 weeks after changing. For warfarin, expect more frequent INR tests for the first month.
- Know your manufacturer. If you’re on a generic, note the pill’s imprint (letters/numbers on the tablet). If it changes, ask why. Different manufacturers = different formulations.
Pharmacists surveyed in 2022 said 87% believe generic NTI drugs are just as effective as brands. But 94% said they’re safe-only if the patient is monitored. That’s the key: safety isn’t automatic. It’s managed.
State Laws and Insurance Rules
Twenty-eight states have laws that restrict automatic substitution for NTI drugs. In those places, your pharmacist must contact your doctor before switching. Other states? No such protection. You could get switched without knowing it.
Insurance companies are pushing hard for generics. Many require prior authorization before covering brand-name NTI drugs. Blue Cross Blue Shield of Kansas, for example, makes patients pay the full price difference if they choose brand over generic. That’s a $45 monthly hit for some.
But here’s the loophole: if your doctor writes "dispense as written," insurers usually can’t override it. That’s your legal right. Don’t be afraid to ask.
What Should You Do?
Here’s a simple action plan:
- Find out if you’re on an NTI drug. Ask your doctor or pharmacist. If you take levothyroxine, warfarin, tacrolimus, or antiseizure meds, you likely are.
- Check your pill. Look at the imprint. If it changes, ask if it’s the same manufacturer.
- Ask for "dispense as written." Especially if you’ve had any issues in the past.
- Get blood tested after any switch. Don’t wait for symptoms. TSH, INR, and drug levels need checking 4-8 weeks after a change.
- Don’t panic. Most people switch successfully. But don’t assume it’s risk-free.
The truth? Generic NTI drugs work for most people. But they’re not magic. They’re medicine. And medicine, especially narrow-window drugs, demands attention. Your body doesn’t care about the label. It cares about the dose. And if that dose shifts-even slightly-you might not feel it until it’s too late.
What’s Next?
The FDA is launching a new NTI Drug Registry in 2024 to track real-world outcomes after substitutions. The AHRQ is funding a $2.4 million study tracking 50,000 patients over two years. By 2025, we may have clearer guidelines for each specific NTI drug-not just broad rules.
For now, the message is simple: don’t let cost decide your safety. If you’re stable, stay put. If you need to switch, do it with eyes wide open. Your health isn’t a commodity. It’s a balance. And with NTI drugs, every milligram counts.
Are generic NTI drugs as safe as brand-name ones?
For most patients, yes-but not always. Studies show generic levothyroxine and warfarin work just as well as brand names in large populations. But for drugs like tacrolimus and phenytoin, switching between manufacturers can cause dangerous fluctuations in blood levels. The key is consistency: once you’re stable on a specific product (brand or generic), stay on it. Don’t switch unless necessary, and always get blood tests after any change.
Can my pharmacist switch my NTI drug without telling me?
In most states, yes. Pharmacy laws allow automatic substitution for AB-rated generics. But 28 states have laws requiring pharmacist-physician consultation or patient consent for NTI drugs. If you’re on one of these medications, ask your doctor to write "dispense as written" on your prescription. That legally blocks automatic substitution.
Why does my doctor say not to switch my levothyroxine?
Even small changes in levothyroxine absorption can throw off your thyroid hormone levels. A 10% difference in dose can mean the difference between feeling fine and developing symptoms like fatigue, weight gain, or heart palpitations. Studies show about 30% of patients need a dose adjustment after switching brands or generics. Your doctor isn’t being overly cautious-they’re protecting your stability.
How do I know if my generic NTI drug is from a different manufacturer?
Look at the imprint on the pill. The letters or numbers printed on it change between manufacturers. If your pills suddenly look different, ask your pharmacist if it’s the same company. Even if it’s still labeled "generic," a new manufacturer means a new formulation. That’s when you should get a blood test and talk to your doctor.
Should I avoid generics if I’m on a blood thinner like warfarin?
No-you don’t need to avoid generics entirely. Most warfarin users switch successfully. But INR levels can fluctuate during the first few weeks after a switch. That’s why your doctor will likely check your INR more often right after the change. If you’ve had unstable INR readings in the past, ask your doctor to write "dispense as written." Otherwise, monitor closely and don’t skip your blood tests.
William Minks
March 8, 2026 AT 07:34Just switched my levothyroxine to generic last month and didn’t think twice. TSH came back perfect. 🤷♂️ But I’m young, healthy, and don’t have other meds messing with absorption. YMMV. 😊
Jeff Mirisola
March 9, 2026 AT 09:20People act like generics are some kind of scam, but the data doesn’t back that up. FDA’s scaled bioequivalence for NTI drugs is legit. I’ve seen too many patients stress over $60 pills when $5 works just fine. Don’t fear the pill-fear the lack of monitoring. 🙌
Susan Purney Mark
March 10, 2026 AT 01:00My mom’s been on warfarin for 12 years. Switched from brand to generic last year after her insurance dropped coverage. She was terrified. We did INR checks every week for a month. Stable as a rock. 💙
But I get it-change is scary when your life depends on a number. Talk to your doc. Get tested. Stay calm. You got this.
Ian Kiplagat
March 10, 2026 AT 06:04AB rating ≠ identical. Two generics can differ in fillers, dissolution rate, coating. For tacrolimus? Big red flag. My transplant team won’t touch switches. Ever. Simple as that.
Amina Aminkhuslen
March 10, 2026 AT 12:17Oh sweet mercy, here we go again with the ‘trust the FDA’ fairy tales. 🤮
Let me guess-you’ve never had a seizure because some pharmacist swapped your phenytoin for a ‘generic’ that tastes like chalk and works like a wet sock? No? Good. Now go tell that to the guy whose kid had a stroke because his tacrolimus dipped below 3 ng/mL. Real people. Real consequences. Not your lab rats.
amber carrillo
March 11, 2026 AT 21:23Consistency is key. If you’re stable stay put. If you switch monitor closely. Simple advice. Effective. Practical. Done.
Adebayo Muhammad
March 12, 2026 AT 18:49Let’s be brutally honest: the pharmaceutical industry doesn’t care about your thyroid-it cares about your wallet. Brand-name NTI drugs are a monopoly disguised as science. The FDA’s ‘scaled’ bioequivalence? A bureaucratic smokescreen. You think they test on elderly diabetics with renal impairment? No. They test on 22-year-old college kids who drink energy drinks and do push-ups before blood draws. That’s not science. That’s theater. And you’re the audience paying for the tickets.
Meanwhile, 42% of epilepsy patients report seizures after switching? That’s not ‘anecdotal’-that’s a systemic failure. But hey, let’s all just ‘monitor closely’ and pretend this isn’t a profit-driven nightmare. The real question isn’t whether generics work-it’s why we’re forced to gamble with lives to save $45/month.
And before you say ‘but the data shows equivalence’-show me the data from real-world patients on Medicaid in rural Alabama. Not the shiny NIH trials. The ones where someone forgets to eat because they’re choosing between insulin and rent. That’s where the real risk lives. Not in the pill. In the system.
So yes, generics can work. But only if you’re lucky. And privileged. And have insurance that doesn’t treat you like a spreadsheet cell.
Ferdinand Aton
March 14, 2026 AT 13:57Wait, so if I’m on brand levothyroxine and feel great, but my pharmacist swaps it for generic without telling me… and I start feeling tired… and I don’t get my TSH checked… and I gain 20 pounds… and then I get diagnosed with hypothyroidism… is that my fault? Or the system’s? 🤔