Fake Generic Drugs: How Counterfeits Enter the Supply Chain
 Feb, 25 2026
Feb, 25 2026
Every year, millions of people take generic drugs because they’re affordable and effective. But what if the pill in your bottle isn’t what it claims to be? Fake generic drugs are a growing global crisis, and they’re slipping into the supply chain in ways most people never see. These aren’t just poor-quality copies-they’re dangerous fakes that can contain no active ingredient, the wrong ingredient, or even toxic substances. And they’re not just a problem in faraway countries. They’re in pharmacies, online stores, and sometimes even in hospitals.
How Fake Drugs Are Made
Counterfeit generic drugs don’t appear out of nowhere. They’re made in hidden labs, often in places with weak regulation-like parts of Southeast Asia, Eastern Europe, and West Africa. These labs don’t need fancy equipment. A basic printer, a chemical supplier, and a box of blister packs are enough. The fake pills are designed to look identical: same color, same shape, same logo. Some even replicate the smell and texture of the real thing. In 2023, TrueMed Inc. found that counterfeiters now achieve 95% visual accuracy using off-the-shelf printing tools. That means even trained pharmacists can miss the difference.
The real danger lies in what’s inside. Instead of the correct active ingredient, fake drugs might contain chalk, sugar, or worse-chemicals like boric acid or rat poison. In 2008, contaminated heparin from China led to 149 deaths in the U.S. because the raw material was adulterated with a cheaper, toxic substitute. Today, counterfeiters are getting smarter. They use chemically similar compounds that mimic the behavior of real drugs, so lab tests might not catch them right away. The World Health Organization reports that 77% of fake drugs detected in legitimate supply chains are oral tablets, especially for high-demand conditions like hypertension, diabetes, malaria, and antibiotics.
How They Get Into the Legitimate Supply Chain
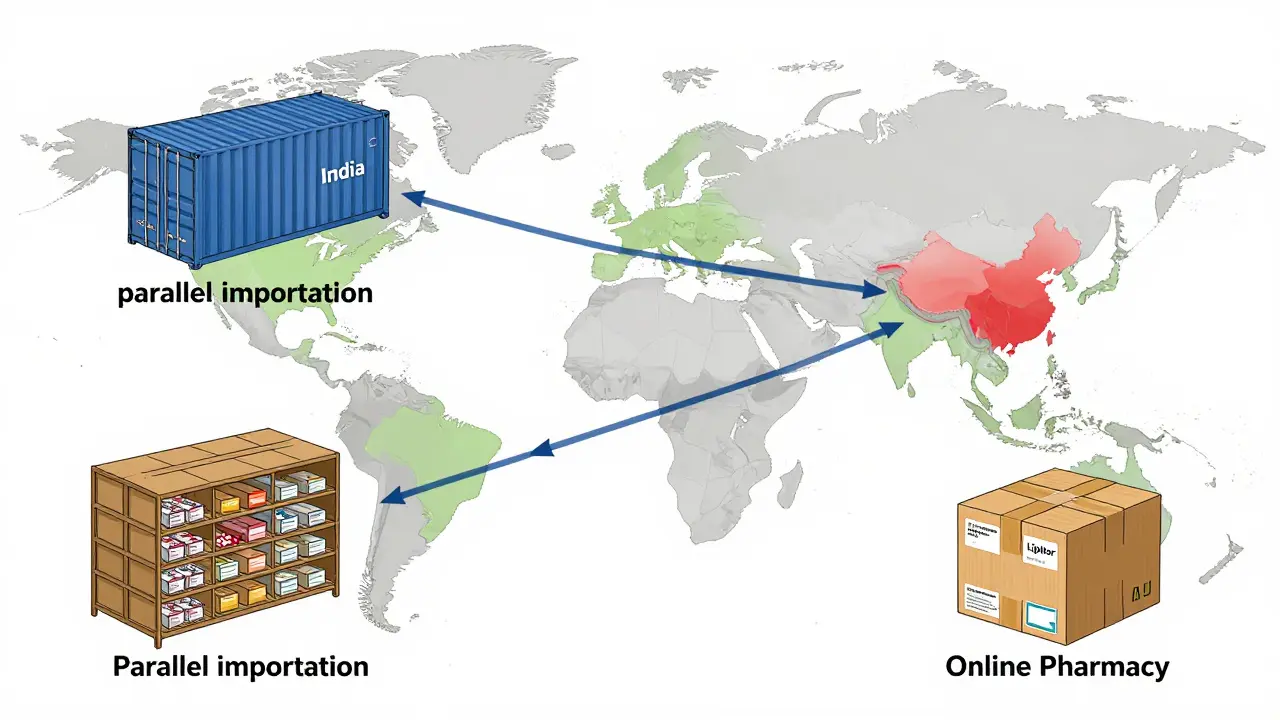
Counterfeit drugs don’t just show up on street corners. They slip into legal distribution networks through three main routes.
First, parallel importation. This happens when drugs bought cheaply in one country are resold in another where prices are higher. Regulatory gaps between countries let fake products ride along with real ones. A batch of fake metformin from India might be mixed in with a legitimate shipment to Nigeria, then resold as genuine.
Second, grey market sales. Unauthorized distributors buy real drugs in bulk, then mix in fake ones to boost profits. These aren’t always shady operations-sometimes they’re small wholesalers trying to cut costs. The International Federation of Pharmaceutical Federations (FIP) found that 68% of pharmacists worldwide have seen suspected counterfeits, but 32% couldn’t tell them apart from the real thing.
Third, online pharmacies. The National Association of Boards of Pharmacy (NABP) says 95% of online pharmacies selling drugs without a prescription are illegal. Many of them operate from hidden servers, often in countries with no enforcement. You might think you’re ordering Lipitor from a U.S. site, but the package came from a warehouse in Bangladesh. Reddit user u/PharmaWatcher reported receiving counterfeit Lipitor with mismatched tablet scoring and color-later confirmed to fail dissolution tests. That’s not a glitch. That’s fraud.
Why Generic Drugs Are Targeted
Counterfeiters don’t go after expensive brand-name drugs like Humira. They go for generics because they’re sold in massive volumes, have thin profit margins, and are less likely to be tracked. The global generic drug market hit $438.7 billion in 2022. That’s a huge target.
Generic manufacturers operate on tight budgets. They’re under pressure to keep prices low, so some cut corners on quality control. This creates openings for counterfeiters to slip in. A 2023 Drug Patent Watch analysis found that the de-formulation process-where generics reverse-engineer brand drugs-can leave gaps in oversight. When a company copies a drug, they’re not always required to prove long-term stability or purity. That’s why impurities like N-nitrosamines kept showing up in blood pressure medications starting in 2018. Counterfeiters exploit the same loopholes.
Where the System Fails
The pharmaceutical supply chain is long. A pill might pass through five or six intermediaries before it reaches you: manufacturer → distributor → wholesaler → pharmacy → patient. Each handoff is a chance for fraud.
Only 40% of countries have full track-and-trace systems. That means there’s no digital record of where each batch went. The U.S. Drug Supply Chain Security Act (DSCSA) required full serialization by 2023, but most developing nations still rely on paper logs. The European Union’s Falsified Medicines Directive cut counterfeit penetration by 18% after requiring tamper-evident packaging and barcode scanning. But in places without those rules, fake drugs flow freely.
Even when systems exist, they’re not foolproof. A 2023 Europol operation seized cancer drugs with AI-generated holograms that passed visual inspections. These weren’t hand-stamped fakes-they were digitally designed to fool scanners. Meanwhile, the World Health Organization estimates that adding security features like DNA tags or color-shifting ink costs $0.02-$0.05 per unit. For low-income countries, that’s a barrier.
What’s Being Done-and What’s Not
Some progress is happening. Pfizer’s anti-counterfeiting program has stopped over 302 million fake doses since 2004 by working with customs, pharmacies, and law enforcement. The Medicrime Convention and TRIPS Agreement require countries to enforce intellectual property laws, but enforcement varies wildly. In the U.S., regulators have tools. In rural India or sub-Saharan Africa, they don’t.
Blockchain pilots like MediLedger are showing promise. In 2022 trials, they detected supply chain anomalies with 97.3% accuracy. But these are still niche. Most pharmacies don’t have the tech or training. The FIP survey showed that most pharmacists need 8-12 hours of specialized training just to spot fakes. That’s not happening.
The most common counterfeits? Cardiovascular drugs (28.7%), antibiotics (22.4%), and antimalarials (18.9%), according to the U.S. Pharmacopeia. In Africa, patients are getting antimalarials with only 10-20% of the needed artemisinin. That doesn’t just fail to cure-it breeds drug-resistant malaria.
What You Can Do
You can’t control the supply chain. But you can protect yourself.
- Buy from licensed pharmacies. Check if your pharmacy is accredited by your national board (like NABP in the U.S.).
- Check the packaging. Look for spelling errors, mismatched fonts, or missing batch numbers. Real drugs have consistent, clean labeling.
- Compare your pills. If the color, shape, or scoring looks different from your last refill, ask your pharmacist.
- Avoid online pharmacies without a physical address or a licensed pharmacist on staff. If it looks too cheap, it’s probably fake.
- Report suspicious drugs. Contact your national drug regulatory agency. One report can trigger an investigation.
Counterfeit drugs are not a distant problem. They’re here. They’re growing. And they’re killing people-quietly, systematically. The system is broken in places, and fixing it will take global coordination, investment, and enforcement. But until then, awareness is your best defense.
How common are fake generic drugs?
The World Health Organization estimates that 1% of medicines in developed countries are counterfeit, but in some low-income regions, that number jumps to 30%. Africa alone accounts for 42% of all falsified medical products globally. Generic drugs are the most common target because they’re high-volume and low-cost, making them easier to replicate and harder to trace.
Can you tell fake drugs apart from real ones by looking at them?
Sometimes, but not always. Many counterfeiters now use advanced printing and packaging to match real products with 95% accuracy. Subtle differences-like slightly off-color tablets, mismatched font sizes, or missing batch codes-can be clues. But without lab testing, even trained pharmacists can miss fakes. That’s why verification systems like barcodes and digital tracking are critical.
Are online pharmacies safe to buy generic drugs from?
Almost all online pharmacies that sell without a prescription are illegal. The National Association of Boards of Pharmacy found that 95% of them operate outside the law. Many are based overseas and ship counterfeit or substandard drugs. Only use online pharmacies that are verified by programs like NABP’s VIPPS or have a physical address, a licensed pharmacist available, and require a valid prescription.
What are the most common fake drugs?
The most commonly counterfeited drugs are those with high demand and low cost: antibiotics (22.4%), cardiovascular medications (28.7%), antimalarials (18.9%), and diabetes treatments. These are often targeted because they’re taken daily, making them reliable for repeat sales. Fake versions may contain no active ingredient, too little, or harmful chemicals.
Why are generics more vulnerable to counterfeiting than brand-name drugs?
Generic drugs are sold in massive volumes at low prices, making them ideal for counterfeiters. Brand-name drugs often have stronger legal protections, patents, and branding security. Generics, on the other hand, rely on simpler packaging and less oversight. With many manufacturers producing the same drug, it’s harder to track every batch. Plus, price pressure leads some legitimate suppliers to cut corners-creating openings for fakes to slip in.
What should I do if I think I’ve been given a fake drug?
Don’t take it. Stop using the medication immediately. Contact your pharmacist or doctor. Report it to your national drug regulatory authority (like the FDA in the U.S. or MHRA in the UK). Save the packaging and any receipts. One report can help authorities trace the source and prevent others from being harmed.
Spenser Bickett
February 27, 2026 AT 06:09my cousin took 'metformin' from a 'pharmacy' in Mexico and ended up in the ER for a week. they told her it was 'generic' but the bottle had no batch number. no one cared.
we need to burn the whole system down. or at least make Big Pharma pay for the fakes.
instead? we get more 'awareness campaigns' and 'report suspicious drugs' like that's gonna stop a guy in Bangladesh printing pills with a $200 printer.
lol.
we're all just one click away from dying because someone decided 'profit margin' is more important than 'not dying'.
Christopher Wiedenhaupt
February 28, 2026 AT 18:36While individual vigilance helps, the real solution lies in mandatory international serialization, transparent supply chain audits, and binding penalties for noncompliance. The EU's FMD model proves regulation works. Why isn't this being scaled globally?
Pharmacists are on the front lines, but they're not equipped with the tools or training to be quality control inspectors. This isn't a consumer problem-it's a policy failure.
John Smith
March 2, 2026 AT 10:42Shalini Gautam
March 2, 2026 AT 11:10But here's the thing-no one in the West wants to admit that their cheap drugs come from the same factories.
We make 40% of the world's generics. We're not the problem. We're the solution that got hijacked.
Stop blaming us. Start fixing the global system that lets middlemen profit off our labor and your suffering.
And yes, I've seen pills with chalk in them. I've also seen real ones made in the same factory. It's not about origin-it's about oversight.
Why don't we fund independent testing labs instead of just pointing fingers?
Steven Pam
March 3, 2026 AT 10:41I started checking my prescriptions last year after reading this. Turns out my last bottle of lisinopril had a slightly different shape. Asked my pharmacist. They checked the batch. It was legit.
But here's the kicker-they didn't even know how to verify it until I asked.
So if you're reading this and you're on meds? Don't just take them. Look at them. Ask questions. Be annoying.
That's how change starts. One suspicious pill at a time.
And yeah, online pharmacies? Avoid them like the plague. I learned that the hard way.
Stay safe out there. We got this.
Timothy Haroutunian
March 4, 2026 AT 01:59We have a healthcare system that's more about billing codes than healing.
And now this? Fake drugs? Of course.
The FDA doesn't even have enough inspectors to check the ones that are supposed to be real.
And yet somehow, we still have people screaming about 'socialized medicine' like it's the end of the world.
Meanwhile, people are dying because a guy in a warehouse in Pakistan mixed sugar with 'antibiotics' and shipped it to a distributor who didn't check because the price was too good.
It's not a conspiracy. It's just capitalism.
And we're all paying for it.
Erin Pinheiro
March 4, 2026 AT 06:48People think they're getting something different but it's usually just a different manufacturer.
Like my last refill of atorvastatin? Looked totally different. I freaked out. Called the doctor. Turns out it was just a different generic brand.
So before you panic about fake pills? Maybe check if it's actually fake or just… different.
Also why is everyone so scared of generics? They're not magic. They're just cheaper.
And yes, I know about the bad cases. But don't turn every pill into a horror story.
tia novialiswati
March 4, 2026 AT 10:48I started checking my meds after my grandma got sick from a fake antihypertensive.
Now I always look at the batch number, the imprint, the color-sometimes I even call the manufacturer.
It's a pain? Yes.
But it's worth it.
And if you're buying online? Stick to VIPPS-certified sites. I've saved so many friends just by saying 'wait, let me check this first'.
You're stronger than you think. Keep asking questions. 💪❤️
Lillian Knezek
March 6, 2026 AT 07:06They want you sick so you keep buying.
Did you know the FDA approves drugs based on 30-day trials?
They don't test long-term effects.
And the 'counterfeit' drugs? They're actually the REAL ones. The brand-name ones are the fakes.
Think about it. Why do they make you pay $200 for a pill when the same chemical costs $0.02 to make?
They're not selling medicine. They're selling control.
And now they're using fake drugs to scare you into buying more expensive 'verified' ones.
It's a trap.
Trust no one. Not even the FDA.
Maranda Najar
March 7, 2026 AT 07:12We have constructed a world wherein the very essence of healing-the molecular intervention that sustains life-is now subject to the capricious whims of unregulated commerce.
Each pill, once a sacrament of scientific progress, has been reduced to a commodity, a mere artifact of globalized entropy.
And we, the unwitting faithful, consume them with the blind trust of pilgrims who have forgotten the sanctity of the altar.
What does it mean to live in a society where your survival depends on a barcode?
Where your life is measured not by the integrity of your body, but by the fidelity of a label?
It is not a crisis of counterfeit drugs.
It is a crisis of civilization.
Christopher Brown
March 8, 2026 AT 14:35Christina VanOsdol
March 9, 2026 AT 14:35Every time I hear 'generic drug' I think 'I hope this doesn't kill me'.
And don't get me started on the online pharmacies.
I bought 'Adderall' from one once. It was a white oval with no imprint.
Turned out it was just caffeine.
And the website? Had a .com domain.
With a .com domain.
And a 'contact us' page that said 'email us for support'.
There was no phone number.
No physical address.
Just a PayPal button.
And now I'm paranoid about every pill I take.
And I'm not even on meds.
Just imagine if I was.
It's terrifying.
And nobody's doing anything about it.
Alfred Noble
March 10, 2026 AT 15:51Let me tell you-most of the fakes don’t come from overseas labs.
They come from within.
We had a case last year where a warehouse worker swapped out a pallet of metformin with fake ones.
He was just trying to make extra cash.
And no one noticed because the packaging matched.
Our barcode system was outdated.
Our staff hadn’t been trained since 2017.
And our audit? Was done by a third party who didn’t open a single box.
It’s not a conspiracy.
It’s neglect.
And it’s everywhere.
Even in the 'trusted' system.
Matthew Brooker
March 11, 2026 AT 15:25People don’t need more awareness.
They need better systems.
Universal access to verified meds.
Government-backed supply chains.
Training for pharmacists.
Not just 'report suspicious drugs' and hope for the best.
It’s not about being paranoid.
It’s about being smart.
And we can fix this-if we stop blaming individuals and start fixing institutions.
One pill at a time.
One policy at a time.
One community at a time.