Environmental Monitoring: How to Test Facilities for Contamination
 May, 2 2026
May, 2 2026
Imagine a single invisible bacterium hiding on a conveyor belt in your food processing plant. It doesn't look like much, but it could trigger a recall that costs millions and ruins your brand's reputation overnight. This is why environmental monitoring is a systematic approach to detecting and controlling contamination in production facilities through regular sampling and testing. It isn't just about cleaning; it’s about proving that your cleaning works before a customer ever gets sick.
Whether you run a pharmaceutical lab or a ready-to-eat (RTE) food facility, the goal is the same: stop contamination vectors before they touch your product. The U.S. Food and Drug Administration (FDA) and other global regulators don’t just want you to clean-they want data. They want proof that your Hazard Analysis and Critical Control Point (HACCP) systems are actually holding the line against microbial hazards.
Why Environmental Monitoring Matters More Than Ever
You might think your facility is clean because it looks clean. But visual inspection misses what matters most. According to the Centers for Disease Control and Prevention (CDC), effective environmental monitoring can identify contamination sources before they impact products. This is critical because foodborne illness outbreaks cost the U.S. economy $77.7 billion annually, according to USDA Economic Research Service data from 2022. Most of these outbreaks are preventable with proper monitoring.
The regulatory landscape has tightened significantly. In the 1990s, the FDA began establishing environmental sampling protocols as part of HACCP systems. By 2008, the European Medicines Agency (EMA) formalized requirements in Annex 1 of Eudralex Vol. 4. Today, the FDA explicitly states that "environmental monitoring and product testing are examples of steps they may take to verify control of microbial hazards." If you aren't monitoring, you aren't verifying. And if you aren't verifying, you're operating on hope, not science.
Understanding Zone Classification: The Foundation of Your Program
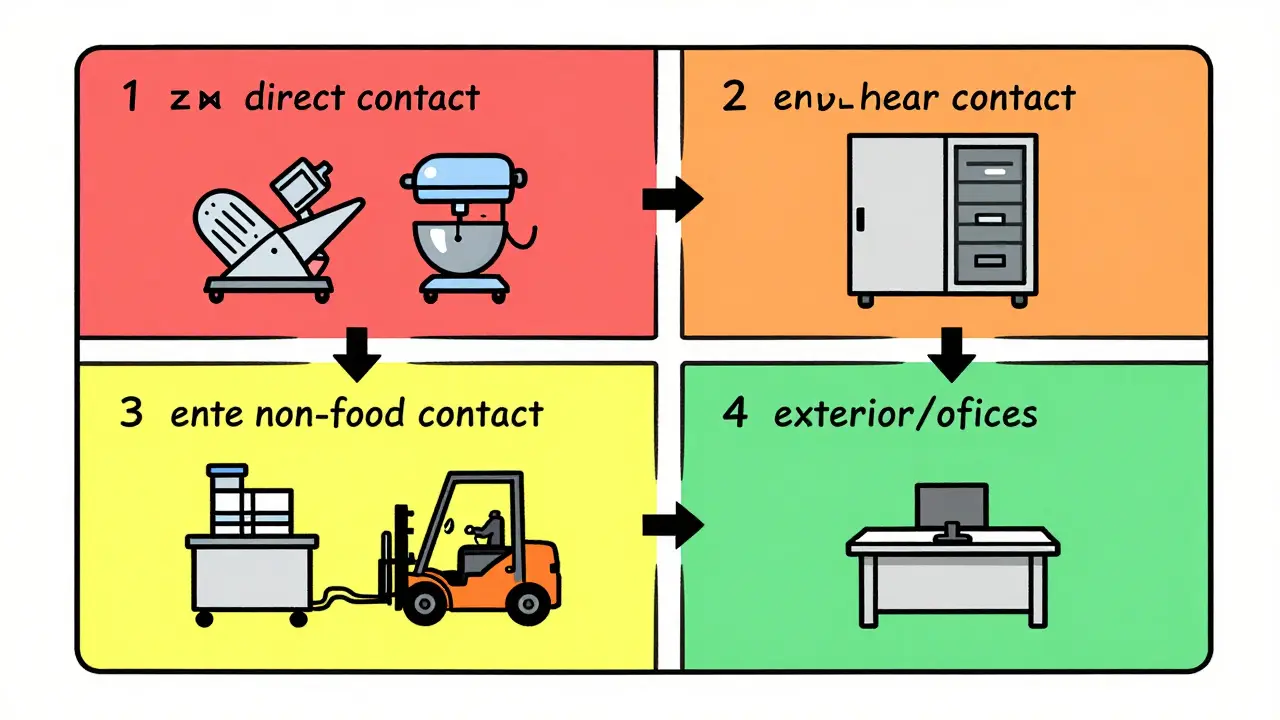
Not all surfaces are created equal. Trying to test every inch of your facility with the same frequency is inefficient and expensive. Instead, successful programs use a zone classification system to prioritize high-risk areas. This framework, detailed in the 3M Environmental Monitoring Handbook published by the International Dairy Foods Association (IDFA), divides your facility into four distinct zones based on risk.
| Zone | Description | Examples | Sampling Frequency |
|---|---|---|---|
| Zone 1 | Exposed food contact surfaces | Slicers, mixers, conveyors, utensils | Daily to Weekly |
| Zone 2 | Non-food contact surfaces near food | Equipment exteriors, refrigeration units | Weekly to Monthly |
| Zone 3 | Remote non-food contact surfaces | Forklifts, remote walls, drains | Monthly to Quarterly |
| Zone 4 | Non-food contact surfaces outside processing | Offices, locker rooms, exterior grounds | Quarterly or As Needed |
Dr. Laurel Dunn of the University of Georgia Cooperative Extension emphasizes that the greatest time and resources should be allocated toward monitoring and cleaning Zone 1 surfaces, followed by Zone 2. However, don't ignore Zones 3 and 4 entirely. A study by PPD Laboratories found that laboratory floors were the source of 62% of all alert and action limit events. Those floors were technically lower-risk zones, yet they harbored significant contamination. Consistent zone classification is key-management at one facility might treat overhead pipes as Zone 1 due to condensation risks, while another treats them as Zone 3. You need a clear, documented policy.
What Are You Actually Testing For?
Your testing strategy depends on what you make. Pharmaceutical facilities often focus on air particulates and specific chemical compounds, while food processors hunt for pathogens. Here’s a breakdown of common analytes and methodologies:
- Microorganisms: General bacteria, specific pathogens (like Listeria monocytogenes or Salmonella spp.), and molds/yeasts. Detected via microbiological methodologies.
- Air-borne Particulates: Measured using liquid impinger or solid impactor samplers. Results are expressed as organisms or particles per cubic meter of air (CFU/m³). Pharmaceutical cleanrooms require continuous non-viable particle monitoring at ISO Class 5 standards.
- Water Quality: Assessed using Total Organic Carbon (TOC) and conductivity measurements. Pharmaceutical facilities must meet USP <645> standards for purified water systems.
- Metals: Detected using Inductively Coupled Plasma (ICP) methodologies.
- Chemical Compounds: Identified using chromatographic methods like GC, HPLC, UPLC, or IC.
For RTE food facilities, the primary targets are almost always Listeria spp. and Salmonella spp.. The USDA Food Safety and Inspection Service (FSIS) requires aggressive Zone 1-4 testing for Listeria under the "Listeria Rule" (9 CFR part 430). If you’re making cheese, deli meats, or fresh produce, this isn't optional-it's mandatory.
Sampling Techniques: Doing It Right the First Time
You can have the best plan in the world, but if your sampling technique is flawed, your data is useless. The CDC notes that many facilities struggle with proper technique, specifically failing to sterilize the interior of sampler devices, which leads to inadvertent cross-contamination.
Here’s how to avoid common pitfalls:
- Use the Right Swabs: For surface sampling, sterile sponges or swabs are standard. Ensure they are validated for the specific pathogen you’re targeting.
- Air Sampling Protocols: Liquid impingers and solid impactors are practical for sampling large volumes of air quickly. Solid impactors come in "slit" or "sieve" designs and must be sterilized before each use.
- ATP Testing for Speed: Adenosine triphosphate (ATP) testing provides results in seconds, compared to 24-72 hours for traditional microbiological tests. The FDA reports that facilities using ATP for sanitation verification achieve 32% faster turnaround between production runs. Use ATP for immediate feedback, but rely on culture-based methods for definitive pathogen identification.
- Training is Non-Negotiable: The FDA recommends at least 40 hours of hands-on training for personnel conducting official monitoring activities. Don’t let someone learn on the job with live production at stake.
Industry Differences: Pharma vs. Food
Your industry dictates your intensity. Pharmaceutical facilities following EU GMP Annex 1 implement more rigorous air particulate monitoring than food processors. They monitor temperature and humidity continuously in critical areas like warehouses and packaging environments. Their goal is sterility.
Food facilities, on the other hand, prioritize pathogen detection. While pharma labs might worry about viable contamination rates staying below 0.01% of environmental monitoring events, food plants are fighting biological threats that cause acute illness. The adoption rate reflects this difference: 98% of pharmaceutical manufacturers have formal environmental monitoring programs, compared to 76% of food processing facilities, according to an FDA 2022 survey. Small food facilities (<50 employees) lag even further, with only 48% maintaining fully compliant programs.
Integrating Data: The Next Step
Collecting samples is only half the battle. The real value comes from integrating data. The 3M Environmental Monitoring Handbook warns that ATP, allergen, and microbiological tests are often siloed. When you combine them, you get a complete picture. PPD Laboratories found that integrating environmental monitoring data with culture contamination frequency tracking reduced false positive rates by 27%.
Look ahead to where the industry is going. The FDA’s 2023 draft guidance encourages next-generation sequencing (NGS) and metagenomics. These technologies can reduce pathogen identification time from 48-72 hours to under 24 hours. Additionally, AI-powered analytics are growing rapidly, projected to reach 38% market penetration by 2027. These tools help you spot trends before they become violations.
How often should I test my facility for contamination?
Frequency depends on the zone and risk level. Zone 1 (direct food contact) should be tested daily to weekly. Zone 2 (near food contact) requires weekly to monthly testing. Zones 3 and 4 (remote areas) are typically tested monthly to quarterly. RTE facilities must test for Listeria in Zone 1 areas at least weekly per FDA guidelines.
What is the difference between ATP testing and microbiological testing?
ATP testing measures adenosine triphosphate, a molecule present in all living cells, providing instant results (seconds) to verify sanitation effectiveness. Microbiological testing identifies specific pathogens like Salmonella or Listeria but takes 24-72 hours. Use ATP for quick checks and microbiological tests for definitive safety verification.
Why are floors considered a high-risk area in environmental monitoring?
Floors are often overlooked as low-risk, but studies show they can be major contamination sources. PPD Laboratories found that floors accounted for 62% of alert and action limit events in bioassay labs. Dust, foot traffic, and moisture can carry pathogens from floors to higher surfaces via air currents or equipment movement.
What are the regulatory requirements for food processing facilities?
Facilities must comply with FDA’s FSMA and USDA’s Listeria Rule (9 CFR part 430). Ready-to-Eat (RTE) facilities face strict scrutiny, requiring aggressive testing for Listeria spp. in Zones 1-4. Documentation of sampling plans, results, and corrective actions is mandatory for inspections.
How much does an effective environmental monitoring program cost?
Medium-sized food processing facilities typically spend $15,000-$25,000 annually on testing supplies and lab services. They also dedicate 2-3 full-time employees to monitoring activities. Costs vary based on facility size, product risk, and whether you outsource lab analysis or perform in-house testing.