Digoxin Generics: Managing Bioavailability and Monitoring Risks
 Apr, 18 2026
Apr, 18 2026
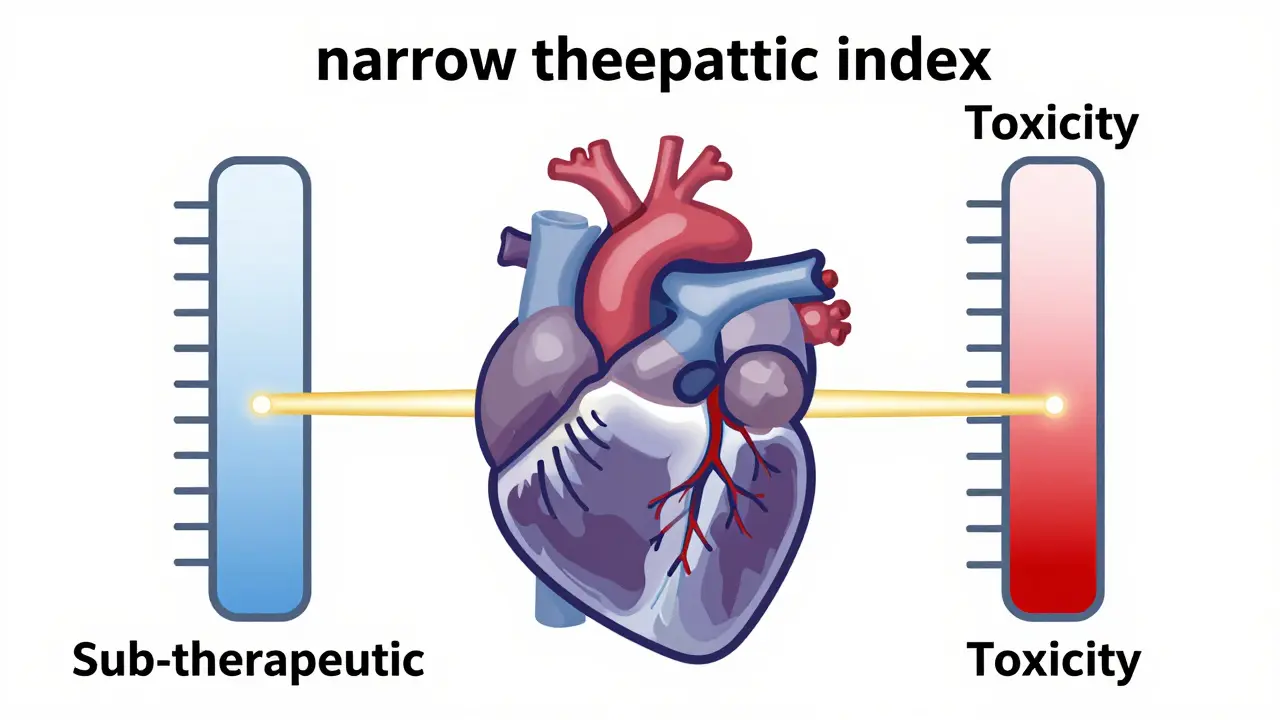
When a pharmacy switches a patient from a brand-name drug to a generic, it's usually a non-event. But for medications with a narrow therapeutic index, a small change in how the body absorbs the drug can be the difference between a stable heart rhythm and a trip to the emergency room. Digoxin is a cardiac glycoside used to treat heart failure and atrial fibrillation by increasing the force of heart contractions and slowing the heart rate. Because the line between a helpful dose and a toxic dose is razor-thin, switching generic manufacturers isn't just a matter of cost-it's a clinical risk.
The Danger of the Narrow Therapeutic Index
Most drugs have a wide safety margin. You can take a slightly higher or lower dose of a vitamin or a common painkiller without much consequence. Digoxin is different. It is classified as a Narrow Therapeutic Index (NTI) drug, meaning its therapeutic range is incredibly tight-typically between 0.5 and 2.0 ng/mL. If the concentration drops slightly below this, the heart failure symptoms return. If it creeps slightly above, the patient risks digoxin toxicity.
This sensitivity makes Bioavailability-the proportion of the drug that reaches the systemic circulation unchanged-critical. Even if two drugs are labeled as the same dose, if one is absorbed at 60% and the other at 45%, the patient's blood levels will shift significantly. For an elderly patient with reduced kidney function, this shift can lead to dangerous drug accumulation.
How the FDA Handles Generic Bioequivalence
To protect patients, the FDA doesn't treat digoxin generics like standard medications. They require an Abbreviated New Drug Application (ANDA) and strict bioequivalence testing. For a generic to be approved, its pharmacokinetic parameters-specifically the Cmax (maximum concentration) and AUC (total drug exposure over 12 hours)-must fall within a 90% confidence interval of 80% to 125% of the reference product, which is Lanoxin.
| Metric | FDA Requirement (Reference: Lanoxin) | Clinical Impact |
|---|---|---|
| AUC [0-12] | 80% - 125% | Total exposure; affects overall drug accumulation. |
| Cmax | 80% - 125% | Peak concentration; relates to immediate toxicity risk. |
| AB Rating | Listed in Orange Book | Indicates demonstrated bioequivalence under FDA rules. |
While these numbers sound safe, there is a catch: these are mean ratios. A study of 12 volunteers might show an average absorption of 85%, which passes the FDA test. However, a specific individual in that group might only absorb 45% of the drug. In a population study, that person is an outlier; in a clinical setting, that person is a patient experiencing therapeutic failure.
The Risk of Switching Between Generics
The real danger often isn't switching from brand-name to generic, but switching from one generic manufacturer to another. While the FDA ensures each generic is bioequivalent to Lanoxin, they don't necessarily require a direct head-to-head bioequivalence study between two different generic versions.
If a patient is stable on "Generic A" and the pharmacy switches them to "Generic B" due to stock issues, the bioavailability difference between those two specific formulations can cause serum concentrations to fluctuate by more than 25%. This is similar to the challenges seen with other NTI drugs, such as tacrolimus, where a change in manufacturer necessitates immediate blood level monitoring to avoid organ rejection or toxicity.
Essential Monitoring Strategies
Because of these variances, Therapeutic Drug Monitoring (TDM) is non-negotiable when changing digoxin products. The goal is to ensure the patient remains within the 0.5-2.0 ng/mL window, though current evidence suggests that for heart failure patients, a tighter target of 0.5-0.9 ng/mL may actually reduce mortality risk.
When a switch occurs, clinicians should follow these steps:
- Establish a Baseline: Measure serum concentration 4 to 7 days after starting therapy or changing a dose.
- Post-Switch Check: Re-check levels 3 to 5 days after switching to a new manufacturer to catch any sudden spikes or drops.
- Trough Timing: Always measure levels as a "trough," meaning just before the next dose is scheduled.
- Symptom Watch: Monitor for toxicity signs like nausea, vomiting, visual disturbances (yellow-green halos), or new arrhythmias.
Formulation Matters: Tablets vs. Elixirs
Not all digoxin deliveries are created equal. The absorption rate varies wildly depending on the form of the medication. For instance, Digoxin Elixir generally shows higher bioavailability-roughly 70% to 85% of an intravenous dose-compared to the tablet form. This means you cannot simply swap a tablet for a liquid version without recalculating the dose and increasing the frequency of monitoring.
Practical Guidelines for Patients and Providers
To minimize the risk of adverse events, the American Heart Association and American College of Cardiology emphasize consistency. If a patient is stable on a specific manufacturer's product, the best course of action is to stick with that exact product. Avoid "pharmacy hopping" or allowing the pharmacy to substitute generic brands without notifying the prescribing physician.
Special attention must be paid to elderly patients. Since digoxin is primarily cleared by the kidneys, any decline in renal function combined with a bioavailability shift creates a dangerous synergy. A generic that is "bioequivalent" for a healthy 25-year-old volunteer in a study may behave very differently in an 80-year-old with chronic kidney disease.
Is a generic digoxin the same as Lanoxin?
On a population level, yes. The FDA requires generics to be bioequivalent to Lanoxin within a range of 80-125%. However, on an individual level, absorption can vary. Because digoxin has a narrow therapeutic index, these small differences can occasionally lead to toxicity or lack of efficacy.
What are the signs of digoxin toxicity?
Common signs include gastrointestinal issues like nausea and vomiting, visual changes (such as seeing blurred lines or yellow-green halos around objects), and heart rhythm irregularities such as bradycardia or arrhythmias.
When should I get my digoxin levels checked?
You should have your levels checked 4 to 7 days after starting the drug or changing the dose. Additionally, testing is required whenever there is a change in renal function, a switch in generic manufacturers, or when starting new medications that might interact with digoxin.
Can I switch between different generic brands of digoxin?
It is generally discouraged. While each generic is compared to the brand-name version, they aren't always compared to each other. Switching between generics can cause serum concentration changes exceeding 25%, which may require a dose adjustment.
Why is the therapeutic range so low for heart failure?
Recent clinical evidence suggests that for patients with heart failure, lower serum concentrations (0.5 to 0.9 ng/mL) are more effective at reducing mortality risk than the higher end of the traditional 0.5 to 2.0 ng/mL range.
Next Steps and Troubleshooting
If you are a provider managing a patient who must switch generic manufacturers, do not assume the dose remains the same. Order a serum digoxin trough level 3 to 5 days after the switch. If the patient is elderly or has stage 3+ chronic kidney disease, increase the frequency of monitoring to once a week until the level stabilizes.
For patients, keep a record of the specific manufacturer of your medication (found on the prescription bottle). If the appearance of the pill changes or the pharmacy mentions a different brand, contact your doctor immediately to arrange for blood work before a potential toxicity event occurs.
Aaron McGrath
April 21, 2026 AT 23:35Absolute madness! We're talking about a razor-thin therapeutic index here. If your Cmax spikes because of a generic swap, you're practically gambling with a cardiac event. Get your TDM sorted or get out of the way! This is basic pharmacokinetics 101 and yet some clinics still treat this like they're dispensing candy.
dallia alaba
April 23, 2026 AT 05:35The point about the mean ratios in FDA testing is a crucial distinction. A population average can look great on paper while leaving a significant percentage of individual patients in the danger zone. It's a systemic gap in how we view bioequivalence.
julya tassi
April 24, 2026 AT 22:59The yellow-green halos part is so spooky! 😵 I had no idea toxicity showed up as visual changes. Definitely makes me want to double check my meds! 😊
Lesley Wimbush
April 24, 2026 AT 23:41Honestly, it's almost quaint that some people still rely on these basic generic standards without demanding a more bespoke approach to their healthcare. Only a truly sophisticated patient would even think to track the manufacturer, but then again, most people just follow the herd. It's just so tragic how the average person navigates the pharmacy counter with zero clue about systemic circulation or AUC. I suppose we can't all be enlightened about the nuances of cardiac glycosides. It's simply an unfortunate reality of the modern medical industrial complex where convenience trumps precision. Just a bit a shimmering example of the mediocrity we accept daily. Truly an eyesore of a system.
Olushola Adedoyin
April 25, 2026 AT 00:57Big Pharma is just playing games with our hearts! They switch the generics on purpose to keep us in a loop of blood tests and doctor visits. It's a racket to keep the labs paid while we're seeing green halos! Wake up!
Tanya Rogers
April 25, 2026 AT 02:50The obsession with 'population averages' is a convenient fiction used to justify subpar regulatory standards.
Brigid Prosser
April 26, 2026 AT 21:15Right on the money with this. It's a proper nightmare when the pharmacy plays musical chairs with your meds. Keep a sharp eye on those labels, folks, or you'll be in a right mess!
Wendy Ajurín
April 27, 2026 AT 21:43I agree with the emphasis on the 0.5-0.9 ng/mL target for heart failure. The data clearly supports a more conservative approach to dosing to improve long-term outcomes.
Truman Media
April 29, 2026 AT 01:29It is wonderful to see such clear guidance for the elderly. We must care for our elders with great patience and precision. 🌟
Aman Tomar
April 29, 2026 AT 08:49This is so scary... i didnt know that changing the company of the drug could make it toxic. the way it is explaind here is very helpful but it makes me feel anxious for my father who takes this medicine. we must be very carefull with the pharamcy substitutions.
William Young
April 30, 2026 AT 19:43Consistency is indeed the safest path forward here.
Cynthia Didion
May 2, 2026 AT 17:07American standards are the only ones that actually matter.
Venkatesh Venky
May 3, 2026 AT 09:09TDM is the gold standard for managing NTI drugs. Just keep the pharmacokinetic flow steady!