Contamination Controls: Preventing Adulteration in Generic Pharmaceuticals
 Feb, 27 2026
Feb, 27 2026
When you take a generic pill, you expect it to work just like the brand-name version. But behind that simple tablet is a complex battle against invisible threats-dust, microbes, chemical residues-that can turn a safe medicine into a dangerous one. This isn’t science fiction. In 2020, a single contaminant-nitrosamine-showed up in blood pressure meds like Valsartan, triggering recalls across 22 manufacturers and costing over $1.2 billion. That’s not an outlier. In fiscal year 2022, contamination was behind 37.2% of all FDA Warning Letters to drug makers. For generic manufacturers, who operate on razor-thin margins, getting this right isn’t optional-it’s survival.
What Counts as Contamination?
The FDA defines a drug as adulterated if it’s made, packed, or stored under unsanitary conditions that could make it harmful. That sounds broad, but it’s precise in practice. Contamination comes in three main forms:- Chemical: Leftover active ingredients from a previous batch, cleaning solvents, or impurities in raw materials. Even 1 nanogram of a potent drug on a surface can cross-contaminate the next one.
- Microbial: Bacteria, fungi, or endotoxins. These don’t just spoil pills-they can cause infections in patients, especially those with weakened immune systems.
- Physical: Glass shards, metal flakes, or even human hair. These are rare but catastrophic when they happen.
It’s not enough to test the final product. The FDA made it clear in 2021: relying on end-product testing alone is a violation of CGMP rules. You have to stop contamination before it starts.
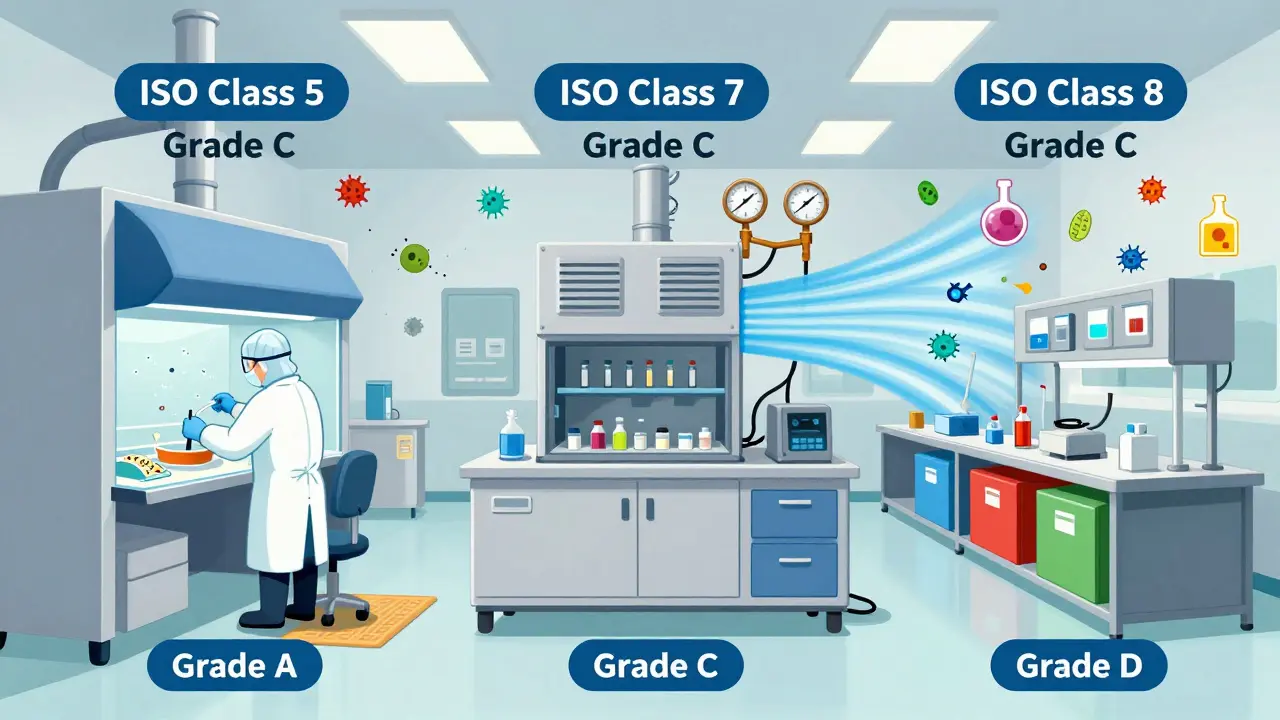
How Clean Is Clean Enough?
Pharmaceutical cleanrooms aren’t like hospital rooms. They’re engineered environments with strict rules. Most generic drug production uses three zones:- ISO Class 5 (Grade A): For filling sterile products. No more than 3,520 particles per cubic meter at 0.5 microns. Think laser-focused air flow, no talking, no sudden movements.
- ISO Class 7 (Grade C): The main production floor. Air changes happen 20-60 times per hour. Pressure is kept 10-15 Pascals higher than adjacent areas to keep dirty air out.
- ISO Class 8 (Grade D): For packaging and storage. Less strict, but still controlled.
It’s not just air. Surfaces must be cleaned to a standard of ≤10 CFU (colony-forming units) per 25 cm². That’s less than one microbe per postage stamp. Chemical residues? No more than 10 ppm of the previous drug. To prove it, manufacturers use swabs, rinse samples, and modern methods like ATP bioluminescence-which gives results in 5 minutes instead of 5 days.
Design Matters More Than You Think
You can’t clean your way out of bad design. Facilities built for contamination control follow three core principles:- Segregation: High-potency drugs (like cancer meds) are made in separate rooms with dedicated HVAC systems. No shared equipment. Ever.
- Unidirectional Flow: Air moves from cleanest to least clean areas. People move the same way-no backtracking.
- Physical Barriers: Airlocks, pass-through hatches, and color-coded equipment prevent mix-ups. One facility reduced errors by 65% just by painting equipment by drug family: blue for blood pressure, red for diabetes, green for antibiotics.
Even small choices matter. Dycem CleanZone mats-sticky, non-woven floor mats-cut foot-borne contamination by 72% in one Pfizer generics facility. That’s not marketing hype. That’s data from their own validation reports.
Human Error Is the Biggest Risk
Technology helps, but people still cause 83% of contamination events, according to industry consultant Dr. Michael Gamlen. Why? Fatigue. Rushing. Poor training.One study found gowning compliance dropped 40% after 8 hours on a 12-hour shift. That’s when gloves tear, masks slip, and people skip steps. Teva had to spend $185,000 upgrading air showers after switching to reusable gowns-because workers weren’t properly decontaminating before entering clean zones.
Training isn’t a one-time event. It’s continuous. Software like ValGenesis V2 can help track cleaning logs, but one survey found it took 147 hours to train a single user. That’s a huge barrier for smaller manufacturers.
Real-Time Monitoring Is Changing the Game
Old-school methods-manual air sampling every few hours-miss 78% of contamination events, according to FDA field data. Why? Contamination spikes in seconds, often from a sneeze, a door opening, or a technician moving too fast.Real-time particle counters like the MetOne 3400+ monitor air continuously. A 2022 ISPE study found they cut contamination incidents by 63%. They’re expensive-$15,000 to $25,000 per unit-but for a facility that’s had even one recall, the ROI is obvious.
Even smarter? AI systems like Honeywell’s Forge Pharma. In a Merck generics pilot, it reduced false alarms by 68% by learning normal patterns. No more waking up at 3 a.m. because a sensor glitched.

The Cost of Doing Nothing
The generic drug market is huge-90% of prescriptions in the U.S. But only 22% of spending. That means manufacturers can’t afford to waste money on over-engineered solutions. But they also can’t afford to cut corners.Some experts warn against “over-engineering.” Dr. Paul Garmory points out that installing ULPA filters (99.999% efficient) for low-risk products like antacids can add $2.8 million a year in energy costs-without meaningful safety gains.
But the cost of failure is worse. A single recall can wipe out a year’s profit. And with FDA increasing inspection frequency by 27% for facilities with past violations, the risk is rising.
What’s Next?
By 2025, the FDA will require all generic manufacturers to use Health-Based Exposure Limits (HBELs) for every product. That means calculating exactly how much of a previous drug can safely remain on equipment. It’s complex. It’s expensive-about $1.2 million per facility to implement. But it’s coming.Meanwhile, innovations are emerging: waterless cleaning systems that cut utility costs by 22%, AI-driven risk prediction models, and continuous manufacturing lines that reduce human contact. The future isn’t just cleaner-it’s smarter.
For generic manufacturers, contamination control isn’t about perfection. It’s about predictability. It’s about knowing, with certainty, that every pill leaving the plant is safe. Because when you’re making medicine for millions, there’s no room for guesswork.
What is the main cause of contamination in generic drug manufacturing?
Human error is the leading cause, accounting for 83% of contamination events according to industry data. This includes improper gowning, cleaning mistakes, and procedural shortcuts-especially during long shifts. Equipment design flaws and raw material issues also contribute, but the biggest risk comes from people under pressure.
How do regulators define a contaminated drug?
Under 21 CFR 210.3(b)(3), a drug is adulterated if it’s prepared, packed, or held under unsanitary conditions that may have caused contamination with filth or rendered it injurious to health. This includes microbial growth, chemical residues above safe limits, or foreign particles. The FDA doesn’t wait for patient harm-any violation of CGMP standards triggers enforcement.
Is cleaning validation still required if I use real-time monitoring?
Yes. Real-time monitoring gives you early warnings, but it doesn’t replace cleaning validation. Validation proves your cleaning process removes residues to acceptable levels (≤10 ppm, ≤10 CFU). Monitoring tells you if that process is working in real time. They’re complementary, not interchangeable.
Why do generic manufacturers struggle more with contamination than brand-name ones?
Brand-name companies often spend $185 million or more on facility design, while generics average $80 million for similar capacity. Generics compensate with tighter operational controls, but they face pressure to cut costs. This leads to compromises in training, equipment, and monitoring-making them more vulnerable to human error and process gaps.
What’s the biggest mistake generic manufacturers make?
Assuming that because a product is low-risk, contamination controls can be scaled back. A 2023 FDA analysis showed even antacids and pain relievers caused recalls when cross-contaminated with potent drugs. Risk-based controls are smart-but underestimating any product’s potential for harm is dangerous.
Can small generic manufacturers afford modern contamination controls?
It’s challenging. Real-time monitoring systems cost $500,000 to $2 million to implement. But the cost of a single recall can be 10x higher. Many small firms are forming cooperatives to share validation labs or adopt phased upgrades-starting with high-risk products first. Delaying investment isn’t saving money-it’s gambling with patient safety.
Katherine Farmer
February 28, 2026 AT 05:19Vikas Meshram
March 1, 2026 AT 21:39Angel Wolfe
March 2, 2026 AT 14:27Charity Hanson
March 2, 2026 AT 16:56bill cook
March 2, 2026 AT 23:58Miranda Anderson
March 4, 2026 AT 23:36Brandon Vasquez
March 6, 2026 AT 05:20Jimmy Quilty
March 8, 2026 AT 04:31Gigi Valdez
March 9, 2026 AT 14:06Byron Duvall
March 10, 2026 AT 04:41Justin Ransburg
March 10, 2026 AT 08:32Sumit Mohan Saxena
March 12, 2026 AT 06:16Sneha Mahapatra
March 12, 2026 AT 09:11Full Scale Webmaster
March 13, 2026 AT 07:20Ben Estella
March 14, 2026 AT 20:50